The Carnot Cycle
The Carnot cycle, proposed by French engineer N. L. Sadi Carnot in 1824, consists of a series of four reversible processes. Because the cycle is entirely reversible, it is the most efficient means of converting heat provided to the system into useful work.
What is Carnot Cycle?
Other examples of thermodynamic cycles are Brayton cycle, Otto cycle, Diesel cycle, Rankine cycle or Stirling cycle etc. All the thermodynamic cycles are the idealized versions of the real processes and are studied to understand the designs of the most efficient systems.
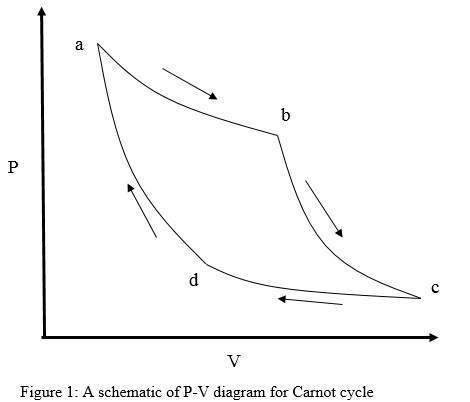
For the Carnot cycle, the four thermodynamic states in the pressure-volume graph are represented by a, b, c and d (Figure 1). Volume, V is plotted on the X-axis while pressure, P is plotted on the Y-axis. The cycle consists of four reversible steps namely
a) Isothermal expansion – With the addition of heat, QH, system expands and goes from state a to b at a constant temperature, TH. Since the process is isothermal, change in internal energy, ΔU is zero.
For the Carnot cycle, the four thermodynamic states in the pressure-volume graph are represented by a, b, c and d (Figure 1). Volume, V is plotted on the X-axis while pressure, P is plotted on the Y-axis. The cycle consists of four reversible steps namely
a) Isothermal expansion – With the addition of heat, QH, system expands and goes from state a to b at a constant temperature, TH. Since the process is isothermal, change in internal energy, ΔU is zero.
b) Adiabatic expansion – System expands from state b to c with no exchange of heat with surroundings while the temperature of the system changes to TC, where TC<TH. As the system is reversibly adiabatic, entropy change, ΔS is zero.
c) Isothermal compression – With the removal of heat, QC, at a constant temperature TC, system compresses from state c to d. Again, since the process is isothermal; change in internal energy, ΔU is zero.
d) Adiabatic compression – System compresses from d to a, with no exchange of heat with surroundings while the temperature of the system increases from TC to TH. Again, ΔS is zero because it is a reversible adiabatic process.
By using ideal gas laws, total work done by the system, w is given by
By using ideal gas laws, total work done by the system, w is given by
(1)
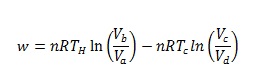
where n and R are the number of moles of gas and the gas constant, respectively. Heat provided to the system, QH is given by
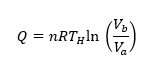
(2)
(3)Efficiency of the Carnot cycle, η is given by
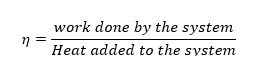
By using (1)-(3) and ideal gas expressions for adiabatic systems, it could be derived that efficiency of a system, η is given by (J.M. Smith, 2005)
(4)
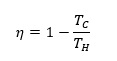
Consequently, larger the difference between hot and cold reservoirs, greater is the efficiency of Carnot cycle. According to the second law of thermodynamics, since all the heat added to the system cannot be converted into work thereby efficiency of any thermodynamic cycle can never be 100%. Also, through (1)-(3) it can be deduced that
(5)
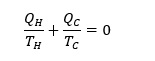
where QC is negative since it is the amount of heat released.
Apart from ideal gas law assumptions, Carnot cycle described above has various other assumptions such as no friction, no heat loss, reversibility of every process, infinite heat reservoir etc. The real processes used in industries have much lower efficiency due to inherent losses of energy in the system. Therefore, ideal Carnot cycle has limited practical applications except for providing the maximum theoretical efficiency achieve by a system.
Nevertheless, it is important to study the cycle. It forms the basis of other thermodynamic cycles that have numerous applications in industries. Two of the applications of the cycle include – Carnot heat engine and Refrigeration.
Carnot heat engine
Carnot heat engine is a hypothetical heat engine designed based on Carnot cycle where heat delivered to a system is used to obtain useful mechanical work. Based on the Carnot cycle discussed above, the heat produced is used to mechanically move a piston which in turn is connected to a crank to produce the motion.
Refrigeration
Refrigeration is a method of removal of heat from one environment at a low temperature and dissipating it in another environment at a higher temperature. As discussed above, it involves four thermodynamic processes that constitute a cycle. Since these processes are reversible, it is possible to go around the cycle in a reverse direction. It consists of four major units namely – a throttling valve, an evaporator, a compressor and a condenser.
A high temperature and a high pressure liquid passes through a throttling valve which reduces its pressure and temperature. The cold liquid is passed through the environment to be refrigerated where it absorbs heat from the environment and changes into vapor. In this process, the circulating fluid absorbs latent heat from the environment to vaporize itself.
After producing the desired cooling effect, the vapor is passed through a compressor where it is compressed into high pressure and high temperature vapor. The last step is converting the vapor back into a liquid, which is done outside the refrigerator where heat is dissipated into surroundings (atmosphere) and the vapor condenses back into liquid.
The high temperature used to calculate maximum Carnot efficiency in this simple refrigeration cycle will be atmospheric temperature. This process removes latent heat of vaporization from the circulating fluid. The cycle is completed after this high pressure liquid is fed to throttling valve. The refrigeration principle is used in industries or in homes to build refrigerators and air conditioners.
J.M. Smith, H. V. (2005). Introduction to Chemical Engineering Thermodynamics. New York: McGraw Hill.
